Boyle’s law 06/07/16
“What is the relationship between Pressure and Volume in a gas?”
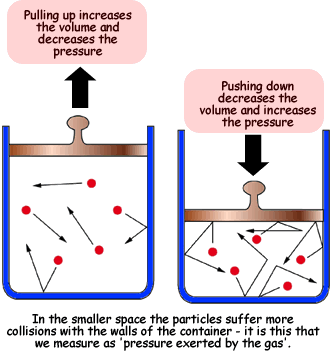
The relationship between Pressure and volume can be neatly summarized in a simple concept known as Boyle’s law. Boyle’s law states that for a gas the pressure multiplied by the gas is simply equal to some constant k. To put this numerically, P*V=k. We can expand upon this by the fact that if the pressure and volume change, their product will be equal to the same multiple, so P1*V1=P2*V2.