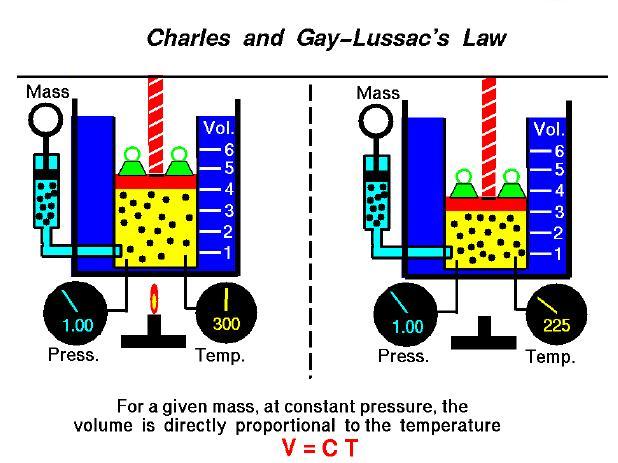
Charle’s law 06/08/16
“ What is the relationship between a gas’ volume and temperature?”
For a gas, the relationship between it’s volume and temperature is very simple. As a gas’s temperature increase, it’s volume will increase proportionally. This is because the molecules of a gas are relatively unbounded to one another, and when their temperature increases, their average speed will increase, and the volume encapsulating the gas will increase as a result. This can be symbolically summarized in Charle’s law, in which VT=k, with Vbeing the volume, T being the temperature, and k being a constant.