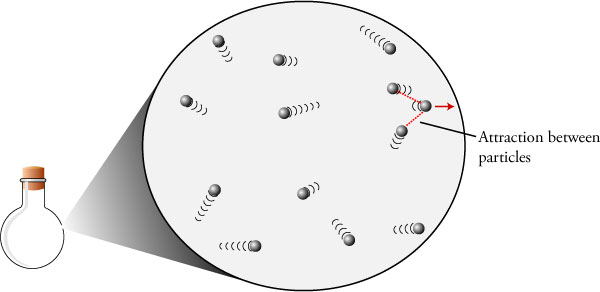
Ideal gas 06/06/16
“What model can we use to describe a gas?”
Let us visualize all of the particles in a gas in their pure intricateness. Think of all of those individuals particles bumping around in semi-random number. There are numerous factors that affect this gas, such as temperature T (how quickly each of these particles are moving around) pressure P(the density of each of the gas particles bouncing against the tank), Volume V (the amount of geometric space the gas takes up), The amount of gas . All of these facets are fundamentally related to each other with something known as the ideal gas law P*V=n*R*T, with R being a constant. This equation is insurmountably amazing because it means we can find out so many properties of a gas by just knowing a few factors!