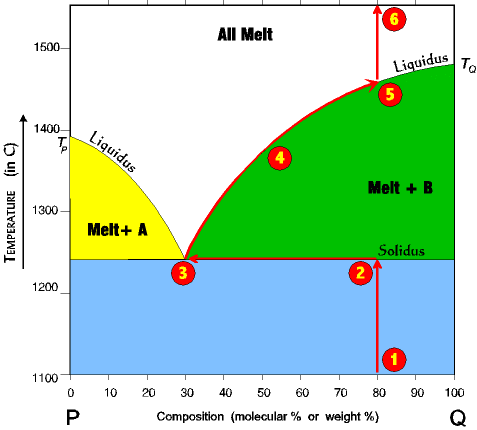
Eutectic point
03/20/17
“What is the lowest possible melting point for a composite material?”
Pure elements always have a fixed melting point. However, when mixed, their melting point can change drastically. Let’s say we have a crystal composed of elements A and B. If element A is predominant, then B will melt first and vice versa for element B. So is there a single point of possible element combinations in which both elements will melt? Well, after years of research, Materials Scientists have came up with the definition of the eutectic point to describe this scenario. The eutectic point is not only the point of complete melting but the point of lowest possible melting since both elements will be melted!